Sterilization Assurance Level
To achieve sterility a device must go through the sterilization process have. An effective process has a very low SAL meaning that there is an extremely small likelihood of any infecting microbes surviving the sterilization.

Pin On 20ways Improving Patient Care Pharmacy Cost Containment Hospital Pharmacy Infusion Pharmacy
While terminal sterilization is conducted on product after it is sealed in its sterile barrier packaging system aseptic processing involves the handling of.

Sterilization assurance level. For sterilization processes the primary device specification is the desired Sterility Assurance Level SAL. Currently a SAL of 10 6 is considered acceptable. Sterility Assurance Level SAL.
Pharmaceutics Abstract Contamination of sterile areas is a concerned issue in pharmaceutical companies it is very hard to overcome these. The major industrial application is in the sterilization of glass bottles which are to be filled aseptically and here the attraction of the process is that it not only achieves an adequate sterility assurance level but that it may also destroy bacterial endotoxins products of Gram-negative bacteria also known as pyrogens that cause fever when injected into the body. Sterilization refers to any process that removes kills or deactivates all forms of life in particular referring to microorganisms such as fungi bacteria spores unicellular eukaryotic organisms such as Plasmodium etc and other biological agents like prions present in a specific surface object or fluid for example food or biological culture media.
In the present context sterilization is the process of removal or inactivation of all microorganisms present on the surface to achieve an acceptable sterility assurance level SAL. How are the Principles of Sterility Assurance Levels SAL Applied to the Sterilization of Liquids by Filtration. 54 provides a sterility assurance level SAL that is possible to calculate validate and control and thus 55 incorporates a safety margin.
This level of probability is significantly greater than that usually attributed to a terminal sterilization process namely 1 in 1 million or 10 6 microbial survivor probability. Sterilization of tissues by ionizing radiation is one of the peaceful applications of nuclear technology for the benefit of mankind. Sterility assurance level or SAL.
Appropriate Sterility Assurance Level SAL for medical devices. It means all 10 6 one million organisms present in the material are killed effectively and assured by extra 6 log reduction. For a healthcare product to be considered sterile the traditional options that have been accessible to a manufacturer are either terminal sterilization to a maximal sterility assurance level SAL of 10 6 or aseptic processing.
Sterility assurance level SAL refers to the probability of a microorganism surviving the sterilization process making the item non-sterile. For aseptic processes a SAL is not applicable as accidental 56 contamination caused by inadequate technique cannot be reliably eliminated by monitor ing control or. The concept of what constitutes sterile is measured as a probability of sterility for each item to be sterilized.
Radiation as a terminal treatment kills or inactivates all the microbes in the processed human tissues to ensure that the tissues attain a high sterility assurance level 10 6 for safe. In practice the sterile state cannot be proven. Which in terms of 10-n the SAL indicates the expected probability of finding a viable microorganism on a medical device.
Sterility Assurance Levels SAL A sterile medical device is free from all viable microorganisms. This probability is referred to as a sterility assurance level SAL. Instead sterility is expressed as the probability of a single viable microorganism occurring on an item after sterilization.
Selection of an appropriate Sterility Assurance Level SAL for medical devices. Appropriate known-to-be-sterile finished articles should be employed periodically as negative controls as a. Sterile hospital instruments must undergo sterilization and SAL.
Assurance Level central services corrosion CSSD HOUSEKEEPING IMPROVEMENT PROCESS Infection INFECTION CONTROL Instrument magic hand of technicians MISSING INSTRUMENTS OPA ophthalmic surgical instruments Ortho-phthalaldehyde Rust SIX SIGMA spot stain surgical instruments Accreditation. A SAL of 106 SAL6 is considered the standard for medical devices and defines the probability of 1 in 1 000 000 that a device is not sterile 108. For each additional two.
Sterility Assurance Level SALs are used to quantify the level or probability of sterility achieved through a certain sterilization process. This probability is commonly referred to as the sterility assurance level SAL of the product and is defined as the probability of a single viable. Read more about the sterility assurance level HERE.
Generally in sterilization it is required to achieve a SAL sterility assurance level of 10 6 and an additional 6 log reduction. Sterility Assurance Level and Aseptic Manufacturing Process in Pharmaceuticals Yasir Mehmood Faculty of Pharmacy University of Central Punjab Lahore Pakistan E-Mail. Whether sterilization takes place using thermal effects heat photo effects UV energy or nuclear effects irradiation each level of increasing efficacy costs energy and well as time.
Free from viable microorganisms. Indicators of health services. The sterility of a product is defined by the probability of a single viable microorganism occurring on an item after sterilization.
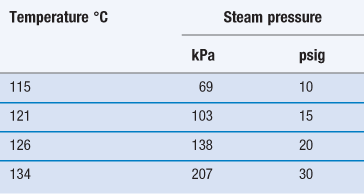
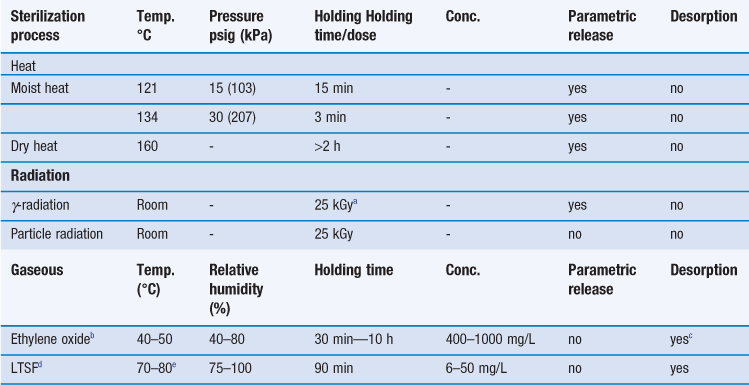
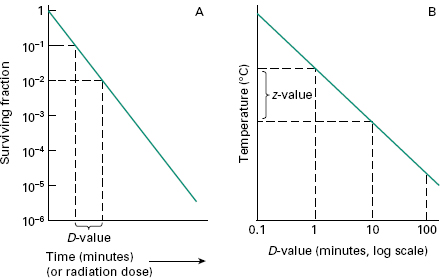
Many factors go into this selection but since the choice of SAL is directly related to sterilization dose this decision is critical to making a sterile claim on any product and one must consider the impact on the product design materials selection customer. This means there is a one in ten thousand 10-4 chance of a single viable Bst spore surviv-ing the process. Steam Sterilization Principles of at least 20 minutes at 121C 250F for a Sterility Assurance Level SAL of 10-4 assuming a starting population of one million 106 organisms.
This TechTip provides information on the selection of an appropriate Sterility Assurance Level SAL for medical devicesMany factors go into this selection but since the choice of SAL is directly related to sterilization dose this decision is critical to making a sterile claim on any product and one must. The current standard for pharmacopoeial sterilization procedures requires a probability of one viable microorganism or less in one million sterilized items of the final product and that means a SAL of 10. Other specifications may include sterilant residues and endotoxin levels.
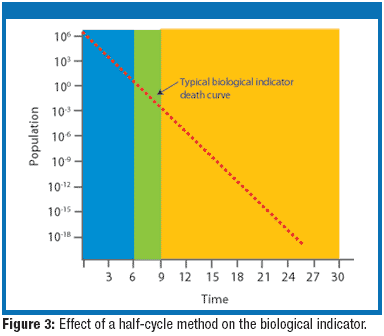
This figure expresses the probability of a single itemin a batch being non-sterile after being subjected to a sterilization process. Sterilization techniques based on bioburden destruction by heat or irradiation require a. Approach to Sterility.

D 10 Values Of Bacterial Isolates And Rsds Required For 10 6 Sterility Download Scientific Diagram

Sterility Assurance Level Fda I3cglobal
Sterilization In Practice Basicmedical Key

Plasma Sterilization What Are The Conditions To Meet This Claim Von Woedtke 2008 Plasma Processes And Polymers Wiley Online Library
Sterilization In Practice Basicmedical Key
Understanding Overkill Sterilization An End To The Confusion

Plasma Sterilization What Are The Conditions To Meet This Claim Von Woedtke 2008 Plasma Processes And Polymers Wiley Online Library
Sterilization Control And Sterility Sterilization Procedures And Sterility Assurance Microbiology

Plasma Sterilization What Are The Conditions To Meet This Claim Von Woedtke 2008 Plasma Processes And Polymers Wiley Online Library

Design Qualification Dq Of Equipment Qualifications Design How To Plan

Rxinsider The Business Of Pharmacy Hospital Pharmacy Patient Care Pharmacy

Simultaneous Achievement Of Sterility Assurance Level Sal Of 10 6 And Material And Functional Compatibility In Gas Plasma Sterilization Running Title Simultaneous Sal And Compatibility Semantic Scholar
Sterilization Procedures And Sterility Assurance Basicmedical Key
Revox Sterilization Solutions Sterility Assurance Level Sal Vs Log Reduction Understanding The Difference Http Bit Ly 2v4pv6t Facebook

Medical Packaging Lessons The 4 Basics Medical Device Companies Should Know And Be Ready For Packaging Medical Packaging Packaging Packaging Design

Sterility Assurance Level An Overview Sciencedirect Topics
Sterilization Procedures And Sterility Assurance Basicmedical Key
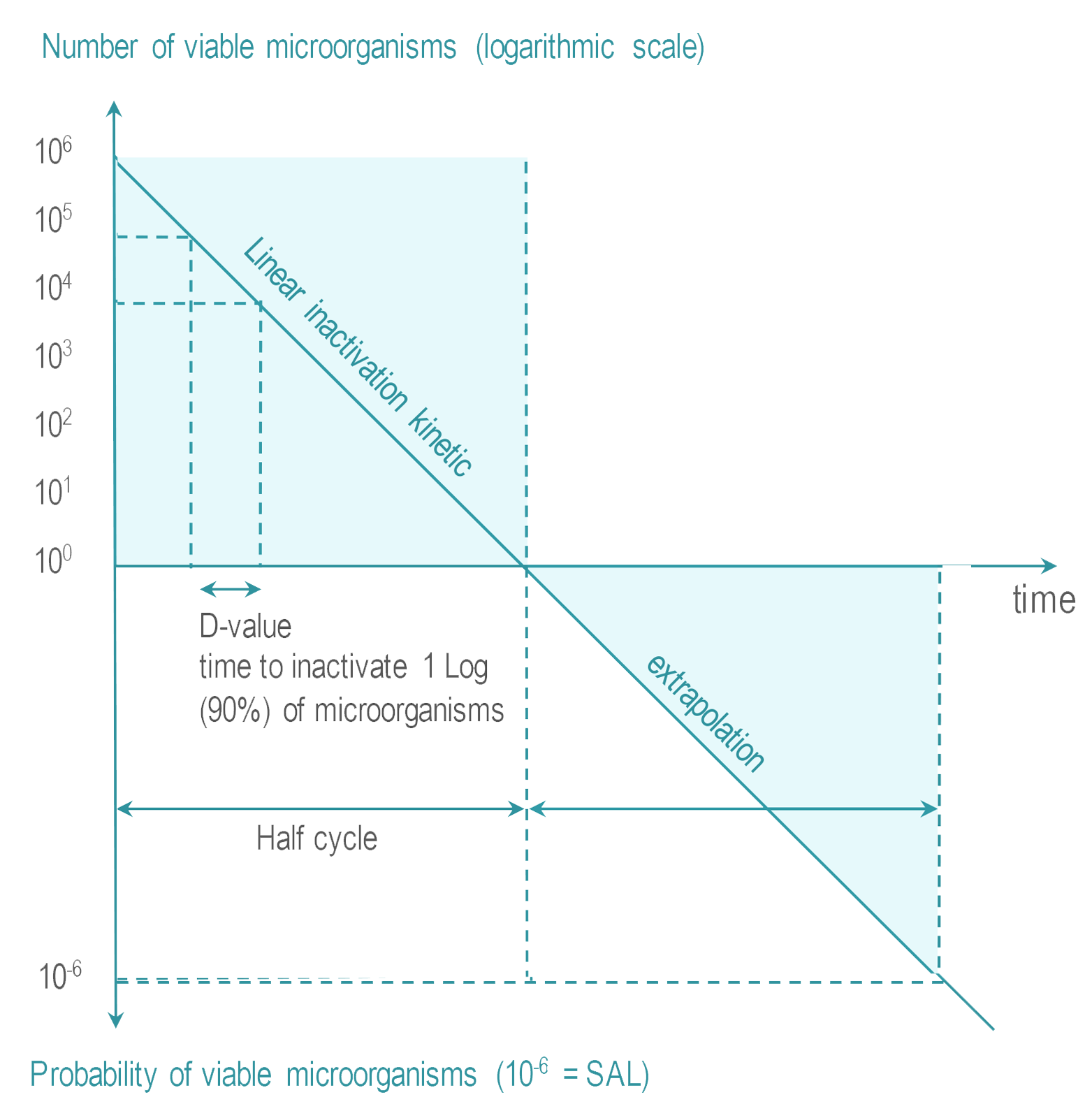
Sterilization Wfhss Guidelines

Posting Komentar untuk "Sterilization Assurance Level"